
Italy Early Toxicity Testing Market by Technique (In Vivo, In Vitro, and In Silico), by Toxicity Endpoint (Genotoxicity, Dermal Toxicity, Skin Toxicity, Ocular Toxicity, Phototoxicity, and Others), by End User (Pharmaceutical Industry, Cosmetic Industry, Chemical Industry, Food Industry, and Others) – Opportunity Analysis and Industry Forecast, 2023-2030
Industry: Healthcare | Publish Date: 03-Dec-2024 | No of Pages: 93 | No. of Tables: 83 | No. of Figures: 32 | Format: PDF | Report Code : HC736
US Tariff Impact on Italy Early Toxicity Testing Market
Trump Tariffs Are Reshaping Global Business
Market Definition
The Italy Early Toxicity Testing Market was valued at USD 46.67 million in 2022 and is predicted to reach USD 100.09 million by 2030, with a CAGR of 8.61% from 2023 to 2030. Early toxicity testing serves as a crucial step in the evaluation of potential drugs, chemicals, or substances, aiming to detect and assess any harmful effects or toxicity during their initial stages of development. This assessment, usually carried out using in vitro methods or animal models, takes place before any experimentation on humans.
The primary goal of early toxicity testing is to promptly identify any safety concerns associated with a substance, enabling well-informed determinations regarding its viability for further advancement and subsequent trials. Engaging in early toxicity testing helps reduce the potential for harmful outcomes in both human participants and animals throughout clinical trials. This not only minimizes costs but also saves time in the broader course of drug development. This complex procedure involves a wide range of analyses and tests, including evaluations of cell viability, assessments of genotoxicity, and investigations into pharmacokinetics.
The specific modalities employed are contingent on the intrinsic nature of the substance under scrutiny and its intended application. Recognized as an indispensable phase within the drug development journey, early toxicity testing profoundly bolsters the assurance of safety and efficacy for novel drugs and other substances, prior to their regulatory endorsement for human utilization.
Expanding Early Toxicity Testing Market in Italy Fuelled by Aging Population's Safety Needs
Growth of the early toxicity testing market in Italy is driven by rise in number of elderly people in the population, who require assurance of drug and chemical safety through early testing to prevent adverse reactions and ensure patient safety. For instance, according to the reports from the World Bank data, the percentage of people above the age of 65 in Italy reached 24% in 2021, as compared to 23% in 2020, and it is expected to increase in the coming years. With rapid growth in ageing population, need for safe medication with minimal or no long-term toxicology is increasing in the Country.
Increasing Prevalence of Chronic Diseases Spurs Demand for Novel Drugs and Therapies in Italy
The rise in prevalence of chronic diseases such as cardiovascular, cancer, and diabetes in the country further propels the demand for new drugs and medication for treatment of these diseases. For instance, according to a report published by the World Health Organization (WHO), the major cause of death in Italy were chronic diseases such as cardiovascular and cancer in 2020, which is driving the demand for early detection and prevention of chronic diseases and emphasizing drug safety. Thus, it is expected to boost the growth of the Italy early toxicity testing market.
The Influence of Regulatory Limitations on the Growth of the Early Toxicity Testing Market in Italy
The stringent regulations set by regulatory bodies such as the US FDA and the EMA require extensive and rigorous testing for drug development and safety, which can be time-consuming and expensive and restrain growth of the market. The complexity and cost of complying with these regulations can pose a significant challenge for small and medium-sized companies that may not have the resources to carry out extensive testing.
As a result, some companies may choose to delay or abandon drug development projects, which can limit demand for early toxicity testing services. Moreover, stringent regulations can also result in a lengthy approval process for new drugs, further delaying the time it takes for drugs to reach the market. This, in turn, is expected to hamper growth of the market.
Technological Advancements in the field of Early Toxicity Testing Market in Italy
Introduction of new technologies such as in-vitro modelling using 3D cell culture is expected to provide new lucrative opportunities for the early toxicity testing market during the forecast period. Use of 3D cell cultures can better mimic complexity of tissues and organs, providing more accurate and reliable results for toxicity testing. Traditional 2D cell culture models are limited in their ability to mimic complexity of human tissues and organs, often leading to inaccurate and unreliable results in toxicity testing.
However, use of 3D cell culture models can better mimic structural and functional complexity of tissues and organs, providing more accurate and reliable results for toxicity testing. 3D cell cultures allow growth and interaction of multiple cell types, creating a microenvironment that more closely resembles human tissues and organs. This can better predict toxic effects of drugs and chemicals in the human body, reducing the risk of adverse effects in clinical trials. Hence, such factors propel the market growth.
Competitive Landscape
The Italy early toxicity testing industry includes several market players such as Inotiv Inc., Bio-Rad Laboratories Inc, Evotec A.G., Agilent Technologies Inc, Wuxi Apptec, Bruker, Perkinelmer Inc., Enzo Biochem Inc., Danaher Corporation, Eurofins Scientific SE, Charles River Laboratories International, Inc., Labcorp Drug Development., Promega Corporation, Insphero AG., and Thermo Fisher Scientific Inc.
Key Benefits
-
The Italy early toxicity testing market report provides a quantitative analysis of the market estimations throughout 2023-2030 that assists in identifying the prevailing market opportunities to capitalize on.
-
The study comprises a deep-dive analysis of the current and future Italy early toxicity testing market trends, depicting the prevalent investment pockets in the industry.
-
The information related to key drivers, restraints, and opportunities and their impact on the market is provided in the report.
-
The competitive analysis of the key players along with their market share in the Italy Early Toxicity Testing industry, is mentioned.
-
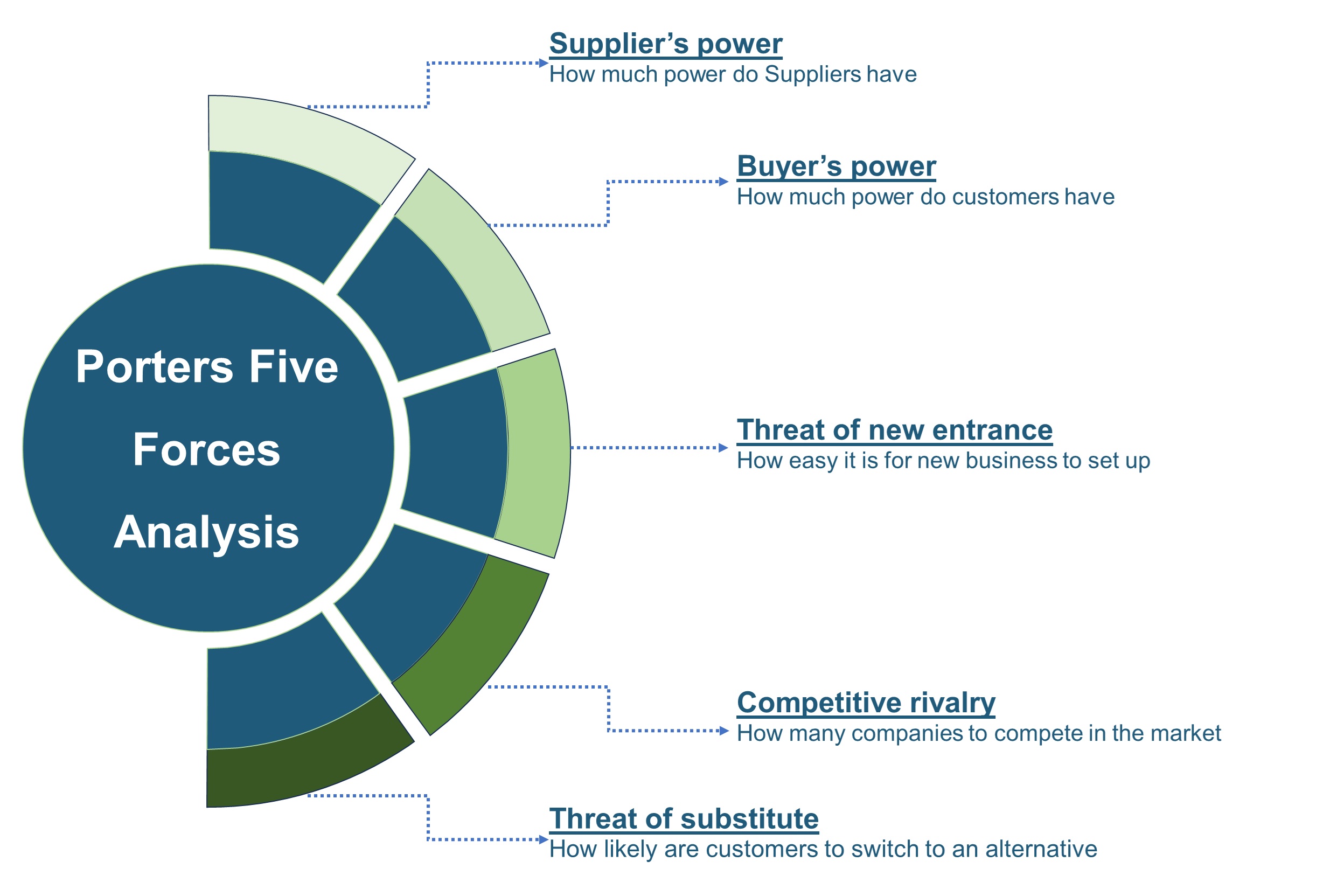
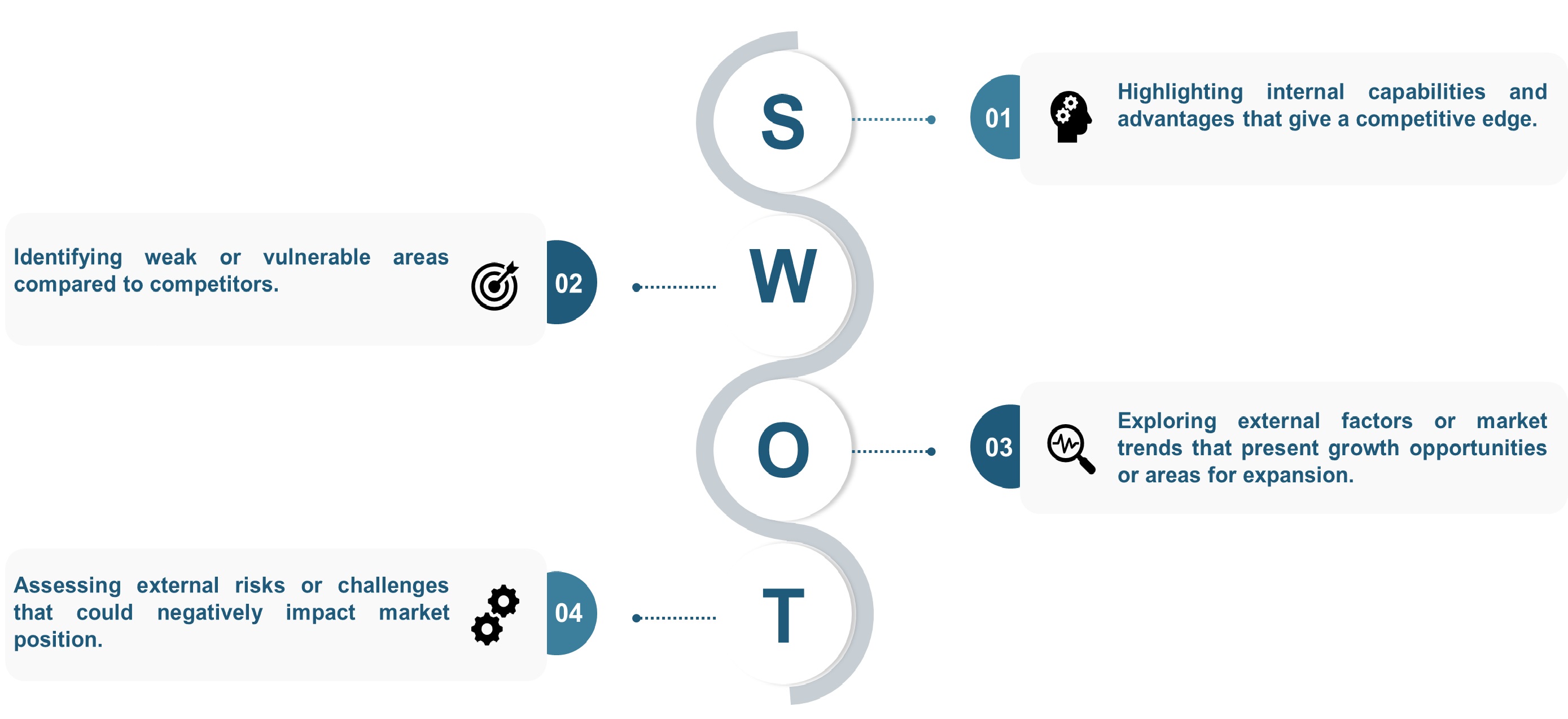
The SWOT analysis and Porter’s Five Forces model are elaborated on in the study.
-
Value chain analysis in the market study provides a clear picture of the stakeholders’ roles.
Italy Early Toxicity Testing Market Key Segments
By Technique
-
In Vivo
-
In Vitro
-
Cell Culture
-
PCR
-
ELISA
-
Western Blotting
-
Protein Binding Assays
-
-
In Silico
By Toxicity Endpoint
-
Genotoxicity
-
Dermal Toxicity
-
Skin Toxicity
-
Ocular Toxicity
-
Phototoxicity
-
Others
By End User
-
Pharmaceutical Industry
-
Cosmetic Industry
-
Chemical Industry
-
Food Industry
-
Others
REPORT SCOPE AND SEGMENTATION:
|
Parameters |
Details |
|
Market Size in 2022 |
USD 46.67 Million |
|
Revenue Forecast in 2030 |
USD 100.09 Million |
|
Growth Rate |
CAGR of 8.61% from 2023 to 2030 |
|
Analysis Period |
2022–2030 |
|
Base Year Considered |
2022 |
|
Forecast Period |
2023–2030 |
|
Market Size Estimation |
Million (USD) |
|
Growth Factors |
The growing incidence of chronic illnesses. Technological advancement. |
|
Companies Profiled |
15 |
|
Market Share |
Available for 15 companies |
|
Customization Scope |
Free customization (equivalent up to 80 working hours of analysts) after purchase. Addition or alteration to country, regional, and segment scope. |
|
Pricing and Purchase Options |
Avail customized purchase options to meet your exact research needs. |
Key Players
-
Inotiv Inc.
-
Bio-Rad Laboratories Inc
-
Evotec A.G.
-
Agilent Technologies Inc
-
Wuxi Apptec
-
Bruker
-
Perkinelmer Inc.
-
Enzo Biochem Inc.
-
Danaher Corporation
-
Eurofins Scientific SE
-
Charles River Laboratories International, Inc.
-
Labcorp Drug Development.
-
Promega Corporation
-
Insphero AG
-
Thermo Fisher Scientific Inc.










 Speak to Our Analyst
Speak to Our Analyst




















