
UK Early Toxicity Testing Market by Technique (In Vivo, In Vitro, and In Silico), by Toxicity Endpoint (Genotoxicity, Dermal Toxicity, Skin Toxicity, Ocular Toxicity, Phototoxicity, and Others), by End User (Pharmaceutical Industry, Cosmetic Industry, Chemical Industry, Food Industry, and Others) – Opportunity Analysis and Industry Forecast, 2023-2030
Industry: Healthcare | Publish Date: 03-Dec-2024 | No of Pages: 93 | No. of Tables: 83 | No. of Figures: 32 | Format: PDF | Report Code : HC733
Market Definition
UK Early Toxicity Testing Market was valued at USD 158.42 million in 2022, and is predicted to reach USD 289.77 million by 2030, with a CAGR of 6.73% from 2023 to 2030.
Early toxicity testing is a core process designed to evaluate the potential toxicity or detrimental impacts arising from drugs, chemicals, or substances in their initial developmental phases. This comprehensive assessment, frequently executed through in vitro studies or animal models, takes place before any human trials.
The primary aim of early toxicity testing is to promptly identify any safety concerns associated with a substance. This, in turn, empowers informed decisions regarding the substance's viability for ongoing development and subsequent testing phases.
Engaging in early toxicity testing serves to mitigate the potential adverse effects on both human subjects and animals during clinical trials. This dual effect not only enhances safety but also contributes to the reduction of expenses and time allocated to the broader trajectory of drug development. This intricate process entails a diverse array of examinations and assays, encompassing evaluations of cell viability, assessments of genotoxicity, and meticulous pharmacokinetic investigations.
The specific modalities employed are contingent on the intrinsic nature of the substance under scrutiny and its intended application. Recognized as an indispensable phase within the drug development journey, early toxicity testing profoundly bolsters the assurance of safety and efficacy for novel drugs and other substances before their regulatory endorsement for human utilization.
Rising UK Elderly Population Drives Market Expansion Due to Chronic Disease Risk
The expanding geriatric population in the UK is emerging as a significant catalyst driving the growth of the early toxicity testing market. This demographic trend is rooted in the increased susceptibility of elderly individuals to develop chronic diseases.
As this older segment of the population is more prone to conditions such as cardiovascular disorders, diabetes, and neurodegenerative ailments, there arises a corresponding need for long-term treatment involving medications.
For instance, according to the Office for National Statistics (ONS), 11 million people in England and Wales are aged 65 years and over, which is expected to grow further in the coming years.
Escalating Healthcare Expenditure Fuels Market Expansion: Advancing Pharmaceutical Facilities and Disease Treatments
The growing healthcare expenditure by the country for the development of highly advanced pharmaceutical facilities and the development of medication and vaccines for the treatment of numerous diseases and viruses further propels the growth of the market.
According to the Office of National Statistics, healthcare expenditure in the UK in 2021 reached USD 300 billion. The significant healthcare expenditure is creating a higher demand for safer and more efficient drug development processes, leading to the widespread adoption of early toxicity testing in drug development to identify potential safety issues at an early stage. Hence, such factors propel the ETT market growth.
Regulatory Constraints and their Impact on Early Toxicity Testing Market Growth in UK
However, stringent regulations set by regulatory bodies such as the US FDA and the EMA require extensive and rigorous testing for drug development and safety, which can be time-consuming and expensive and restrain the growth of the UK early toxicity testing market.
The complexity and cost of complying with these regulations can pose a significant challenge for small and medium-sized companies that may not have the resources to carry out extensive testing.
As a result, some companies may choose to delay or abandon drug development projects, which can limit demand for early toxicity testing services.
Moreover, stringent regulations can also result in a lengthy approval process for new drugs, further delaying the time it takes for drugs to reach the market. This, in turn, is expected to hamper the growth of the market.
Technological Advancements in the field of Early Toxicity Testing Market in UK
The introduction of new technologies such as in-vitro modelling using 3D cell culture is expected to provide new lucrative opportunities for the early toxicity testing market during the forecast period.
Use of 3D cell cultures can better mimic the complexity of tissues and organs, providing more accurate and reliable results for toxicity testing. Traditional 2D cell culture models are limited in their ability to mimic the complexity of human tissues and organs, often leading to inaccurate and unreliable results in toxicity testing.
However, use of 3D cell culture models can better mimic the structural and functional complexity of tissues and organs, providing more accurate and reliable results for toxicity testing.
3D cell cultures allow the growth and interaction of multiple cell types, creating a microenvironment that more closely resembles human tissues and organs. This can better predict the toxic effects of drugs and chemicals in the human body, reducing the risk of adverse effects in clinical trials. Hence, such factors propel the market growth.
Competitive Landscape
The UK Early Toxicity Testing industry includes several market players such as Inotiv Inc., Bio-Rad Laboratories Inc, Evotec A.G., Agilent Technologies Inc, Wuxi Apptec, Bruker, Perkinelmer Inc., Enzo Biochem Inc., Danaher Corporation, Eurofins Scientific SE, Charles River Laboratories International, Inc., Labcorp Drug Development., Promega Corporation, Insphero AG., and Thermo Fisher Scientific Inc.
Key Benefits
-
The UK early toxicity testing market report provides a quantitative analysis of the current market and estimations through 2023-2030 that assists in identifying the prevailing market opportunities to capitalize on.
-
The study comprises a deep dive analysis of the industry trend including the current and future trends for depicting the prevalent investment pockets in the market.
-
The information related to key drivers, restraints, and opportunities and their impact on the market is provided in the report.
-
The competitive analysis of the key players along with their market share in the UK early toxicity testing market.
-
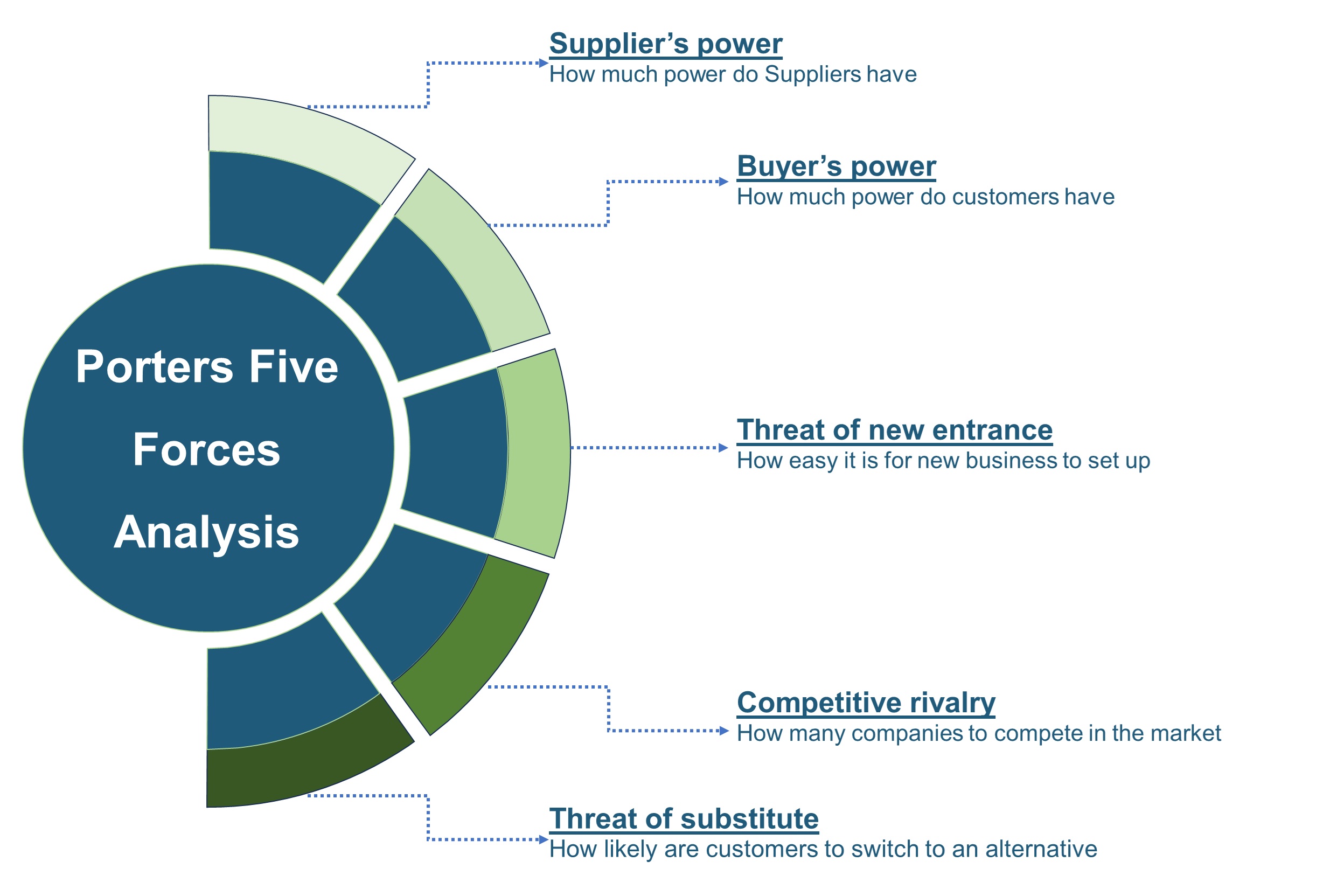
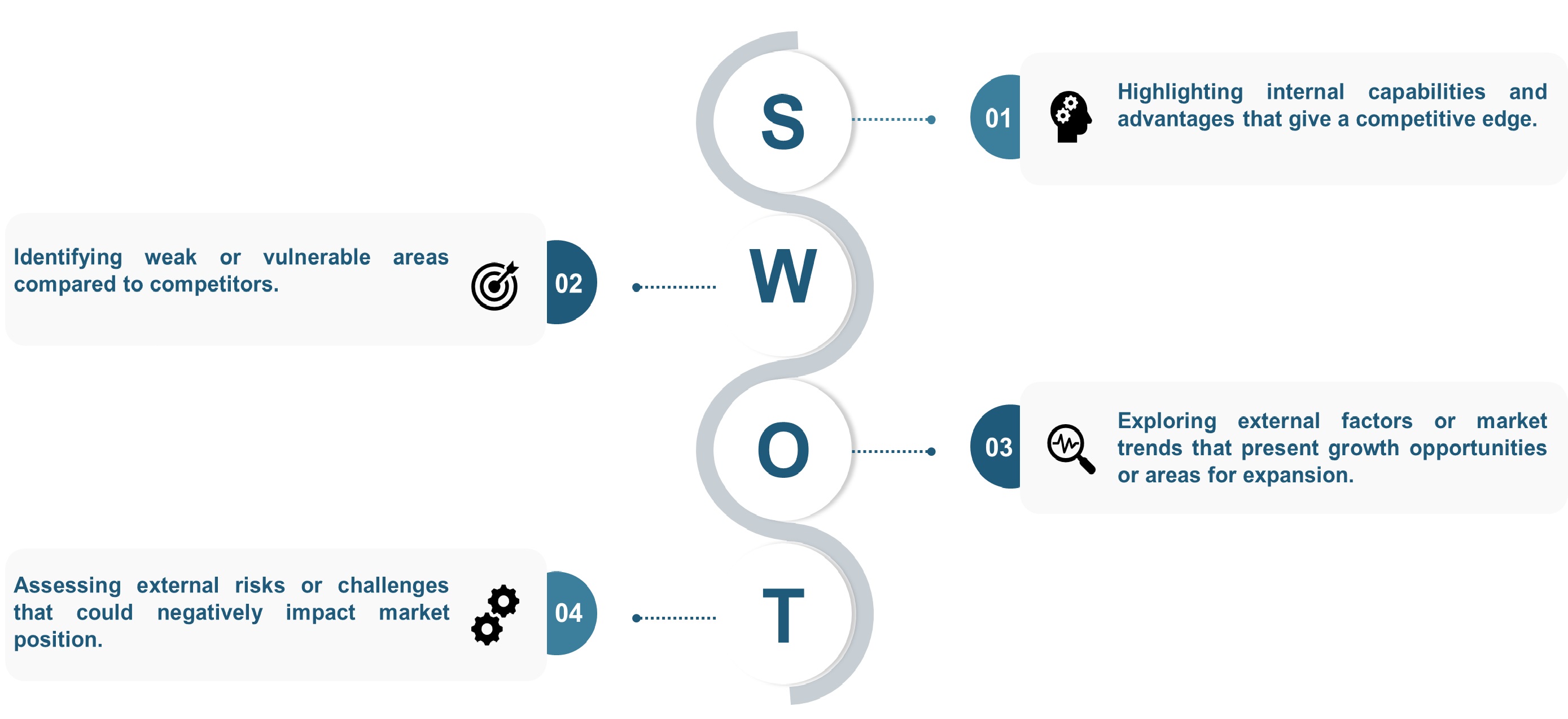
The SWOT analysis and Porter’s Five Forces model are elaborated in the study.
-
Value chain analysis in the market study provides a clear picture of the stakeholders’ roles.
UK Early Toxicity Testing Market Key Segmements
By Technique
-
In Vivo
-
In Vitro
-
Cell Culture
-
PCR
-
ELISA
-
Western Blotting
-
Protein Binding Assays
-
-
In Silico
By Toxicity Endpoint
-
Genotoxicity
-
Dermal Toxicity
-
Skin Toxicity
-
Ocular Toxicity
-
Phototoxicity
-
Others
By End User
-
Pharmaceutical Industry
-
Cosmetic Industry
-
Chemical Industry
-
Food Industry
-
Others
REPORT SCOPE AND SEGMENTATION:
|
Parameters |
Details |
|
Market Size in 2022 |
USD 158.42 Million |
|
Revenue Forecast in 2030 |
USD 289.77 Million |
|
Growth Rate |
CAGR of 6.73% from 2023 to 2030 |
|
Analysis Period |
2022–2030 |
|
Base Year Considered |
2022 |
|
Forecast Period |
2023–2030 |
|
Market Size Estimation |
Million (USD) |
|
Growth Factors |
Rising elderly population, growing healthcare expenditure.
Growing healthcare expenditure. |
|
Companies Profiled |
15 |
|
Market Share |
Available for 15 companies |
|
Customization Scope |
Free customization (equivalent up to 80 working hours of analysts) after purchase. Addition or alteration to country, regional, and segment scope. |
|
Pricing and Purchase Options |
Avail customized purchase options to meet your exact research needs. |
Key Players
-
Inotiv Inc.
-
Bio-Rad Laboratories Inc
-
Evotec A.G.
-
Agilent Technologies Inc
-
Wuxi Apptec
-
Bruker
-
Perkinelmer Inc.
-
Enzo Biochem Inc.
-
Danaher Corporation
-
Eurofins Scientific SE
-
Charles River Laboratories International, Inc.
-
Labcorp Drug Development.
-
Promega Corporation
-
Insphero AG
-
Thermo Fisher Scientific Inc.










 Speak to Our Analyst
Speak to Our Analyst




















